Overexpression of Holocarboxylase Synthetase Predicts Lymph Node Metastasis and Unfavorable Prognosis in Breast Cancer
Witchuda Sukjoi1, Siraprapa Siritutsoontorn1, Pakkanan Chansongkrow1, Suppakit Waiwitlikhit1,2, Steven W. Polyak3, Malee Warnnissorn4, Varodom Charoensawan1,5,6,Chanitra Thuwajit7, Sarawut Jitrapakdee1*
1Department of Biochemistry, Faculty of Science, Mahidol University, Thailand
2Brighton College, Eastern Road, East Sussex, BN2 0AL, United Kingdom
3Clinical and Health Sciences, University of South Australia, 5001, Australia
4Department of Pathology, Faculty of Medicine Siriraj Hospital, Mahidol University,
5Systems Biology of Diseases Research Unit, Faculty of Science, Mahidol University 6Integrative Computational BioScience (ICBS) Center, Mahidol University
7Department of Immunology, Faculty of Medicine Siriraj Hospital, Mahidol University
Source: Anticancer Res 40 (8): 4557-4565.
The biotin-dependent carboxylases (BDCs) are a family of metabolic enzymes that all utilizes biotin as a cofactor to facilitate certain carboxylation reactions. In mammals, BDCs comprise two isoforms of acetyl-CoA carboxylase (ACC-1 and ACC2), methylcrotonyl-CoA carboxylase (MCC), propionyl-CoA carboxylase (PCC) and pyruvate carboxylase (PC), which are intimately involved in various cellular metabolic pathways. Previous studies have reported that BDCs are overexpressed to support growth and survival of several cancers.
Holocarboxylase synthetase (HLCS) is the sole enzyme responsible for attaching the biotin cofactor to a universally conserved lysine present in the active site of all five BDCs Loss of function mutations in human HLCS gene completely abrogate the activity of all five BDCs, reinforcing the critical importance of protein biotinylation in the activation of the BDCs. Although overexpression of the above BDCs has been reported, it is currently unknown whether the levels of HLCS protein are also overexpressed in cancers. Here, we investigate the expression of HLCS protein in breast cancer tissues from 65 Thai patients using immunohistochemistry (IHC).
Materials/methods: The immunohistochemistry was used to investigate HLCS expression in breast tissue obtained from 65 Thai patients, and assessed the correlation between its expression and key clinical-pathological parameters. The role of HLCS in supporting invasion was investigated in HLCS-knockdown MCF-7 cells.
In general, the homogeneous staining pattern of cancer cells in the whole tissue was observed throughout all tissue sections. Weak staining for HLCS expression was observed in the benign mammary glands (Fig. 1A). In contrast, HLCS was highly expressed in the cancerous tissue of stage IV patients (Fig. 1B, same section as Fig. 1A). Notably, no staining was observed in the stromal cells (Fig. 1C, asterisk). An IHC score of 4, representing a median of the total score, was used to classify low (< 4) and high (> 4) expression of HLCS. Based on this criteria, 40 cases (61.5%) had low expression of HLCS while 25 cases (38.5%) showed elevated HLCS expression. In general, the expression levels of HLCS were varied within the same stage i.e. the IHC score varied from 1-7 for stage I, 1-9 for stage II and 0-9 for both stages III and IV. Fig. 1C-1F showed the representatives of IHC of HLCS expression in breast tissue sections of patients with stages I-IV from low to high expression, respectively. Univariate analysis of the IHC showed that expression levels of HLCS were not associated with a specific stage of breast cancer.
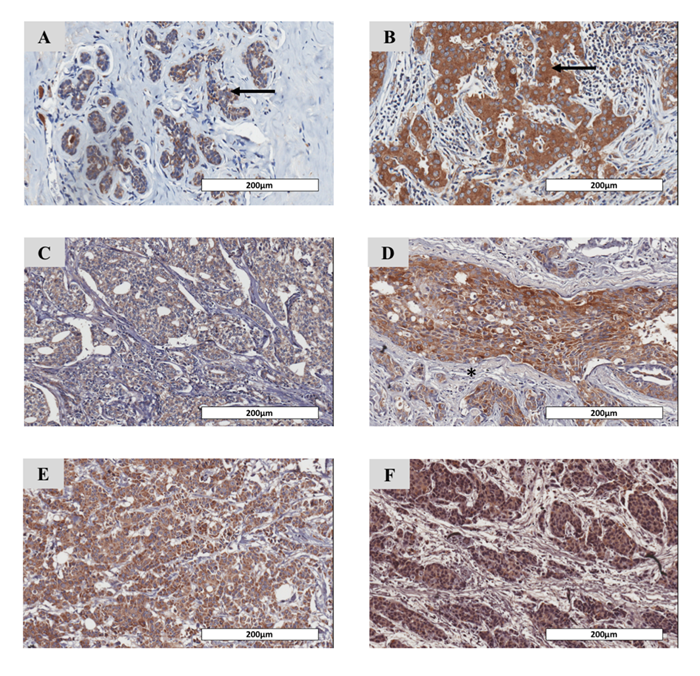
Figure 1. Immunohistochemistry staining of HLCS in paraffin-embedded breast cancer tissue sections. Representative IHC sections showing (A) weak HLCS staining in non-cancerous tissue and (B) strong staining in a cancerous area of stage IV patient. Representatives of IHC staining of breast tissues of patients with stage I-IV (C-F), respectively. Counterstaining of nuclei with hematoxylin is shown in blue. Original magnification was 20x. Scale bar = 200 mm. Arrow represents cytoplasmic staining pattern of HLCS in cancer cells. Asterisk represent no HLCS in stromal cells. HLCS: Holocarboxylase synthetase.
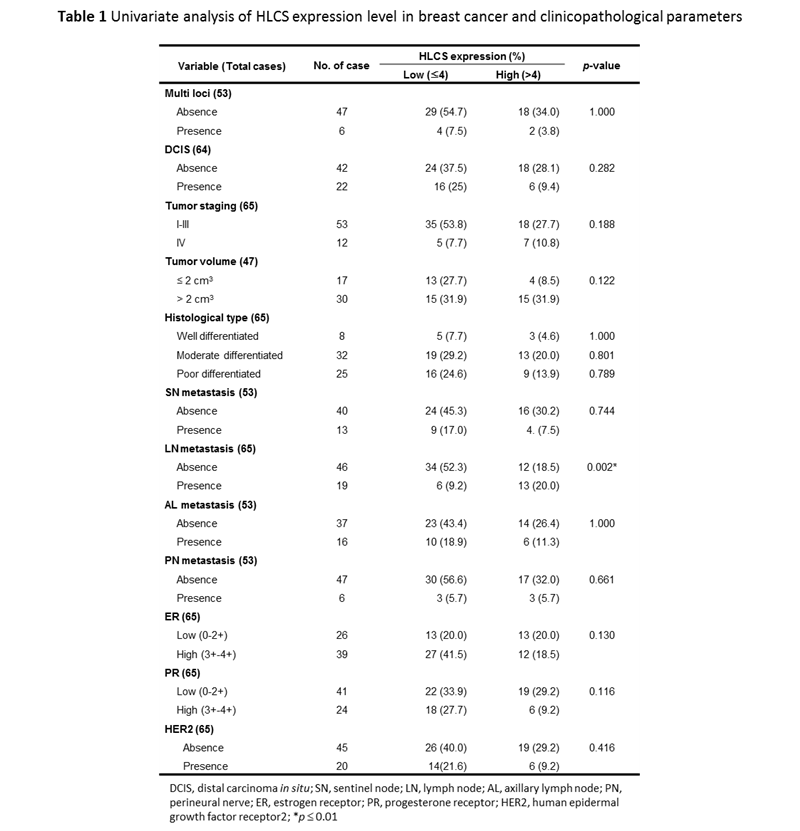
Clinicopathological parameters, including multifoci, ductal carcinoma in situ (DCIS), staging, tumor size, histological grading, lymph node metastasis (sentinel node, SN; axillary lymph node, AL; nodes other than SN and AL, LN; perineural, PN) and biomarkers (estrogen receptor, ER; progesterone receptor, PR; human epidermal growth factor receptor 2, HER2) of all 65 breast cancer patients are summarized in Table 1. Importantly, univariate analysis showed that HLCS over-expression was statistically associated with metastasis of breast cancer to lymph nodes other than sentinel and axillary nodes (13/19 = 68.4%) (p = 0.002). No other correlations were observed for other clinic-pathological parameters (p > 0.05).
In conclusion, the data herein demonstrates the overexpression of HLCS in breast cancer patients in Thailand. A high expression level of HLCS correlated with lymph node invasion and unfavorable prognosis in breast cancer with statistical significance. These results indicate the potentially useful role of HLCS as a prognostic marker for breast cancer. Nevertheless, the biological function of HLCS enzyme in breast cancer should be further analyzed.
| Relevant SDGs | |
|---|---|
 |
|
| BC investigator | |
 Prof.Sarawut Jitrapakdee Prof.Sarawut Jitrapakdee |
 Witchuda Sukjoi(Ph.D. Student) Witchuda Sukjoi(Ph.D. Student) |
